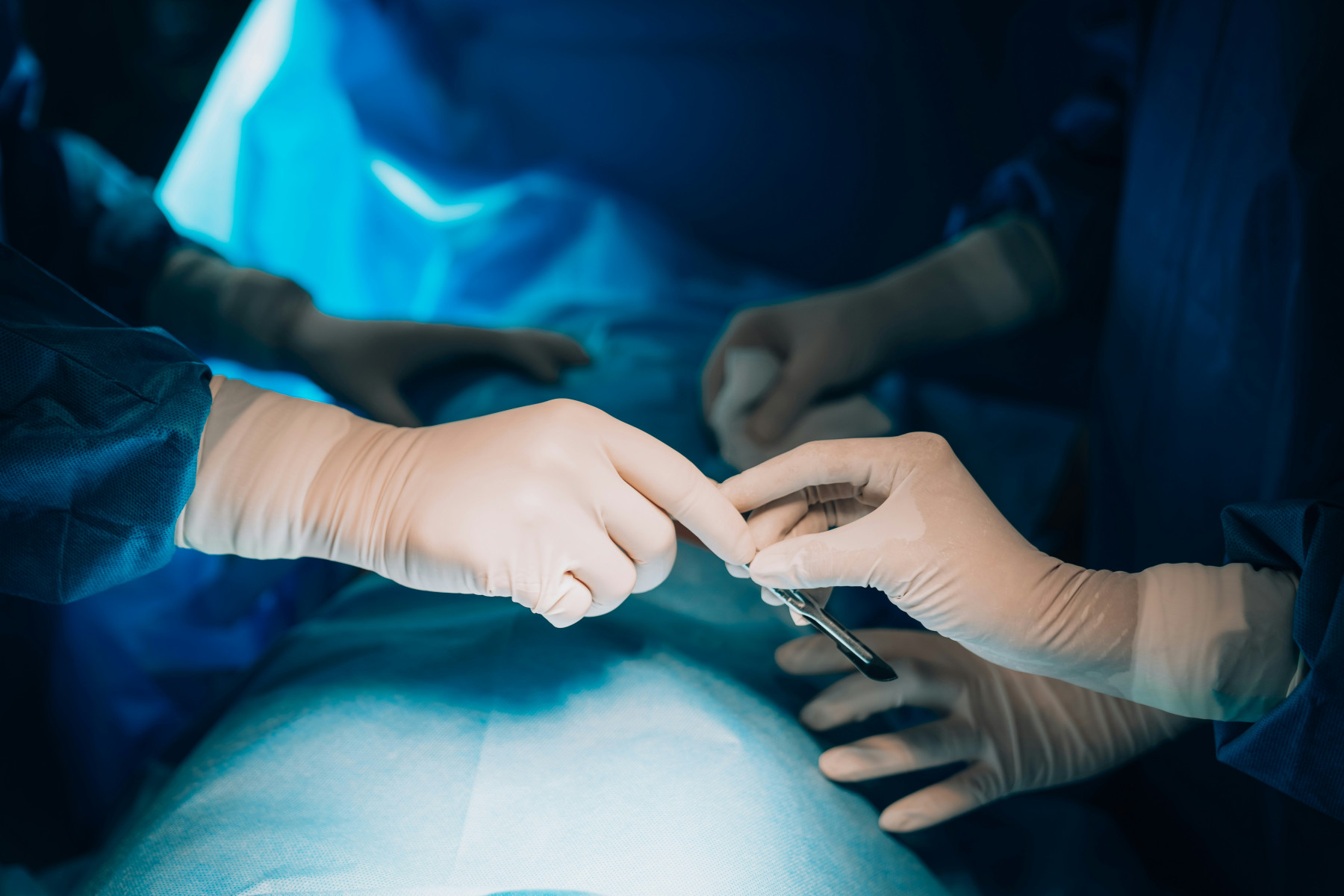
Founded in 1995 in Chivasso (Turin, Italy), Herniamesh S.r.l. designs, manufactures and markets surgical medical devices for inguinal and abdominal hernia repair, as well as for the treatment of female urinary incontinence and pelvic floor disorders. The company's products, covered by proprietary patents, are certified under ISO 13485:2021, bear CE marking and have obtained FDA 510(K) clearance for the US market. Herniamesh distributes its devices internationally through a dedicated commercial network
Vitrex Medical Group (VMG) has acquired Herniamesh S.r.l., a specialist manufacturer of surgical mesh devices for inguinal and abdominal hernia repair and female pelvic health, based in Chivasso (Turin, Italy) and founded in 1995 by Dr. Ermanno Trabucco. The company's CE-marked and FDA-cleared product portfolio is the result of decades of proprietary R&D and patents
VMG is a leading European medical device and IVD manufacturing group, striving to develop, produce and supply high-quality healthcare products and tailormade med-tech solutions. The Group operates through three manufacturing sites located in Denmark, Slovakia and Czech Republic, serving healthcare professionals and patients worldwide
Through this transaction, Herniamesh aims to expand its geographic reach and accelerate its growth by leveraging VMG's international sales network across the USA, Asia and South America. The acquisition represents an important milestone in the development of VMG's CHIRMAX Surgical business unit, enabling the Group to offer a more competitive "Made in Europe" surgical solutions portfolio to hospitals worldwide. The transaction underscores VMG's long-term strategy of combining organic growth with targeted strategic acquisitions to strengthen its position as a global challenger in the surgical med-tech market




















.png)